Determination of the lower explosion point (LEP)
The lower explosion point is the temperature of a flammable liquid at which the concentration of saturated vapor in air equals the lower explosion limit.
The lower explosion point is determined in accordance with DIN EN 15794. The test apparatus used is illustrated in Figure 1. For testing, approx. 120 – 140 g of the product is placed into the glass cylinder (Ø = 88 mm; H = 320 mm). A Huber Ministat 230 thermostat is used to bring the glass cylinder to the proper temperature through a double wall.
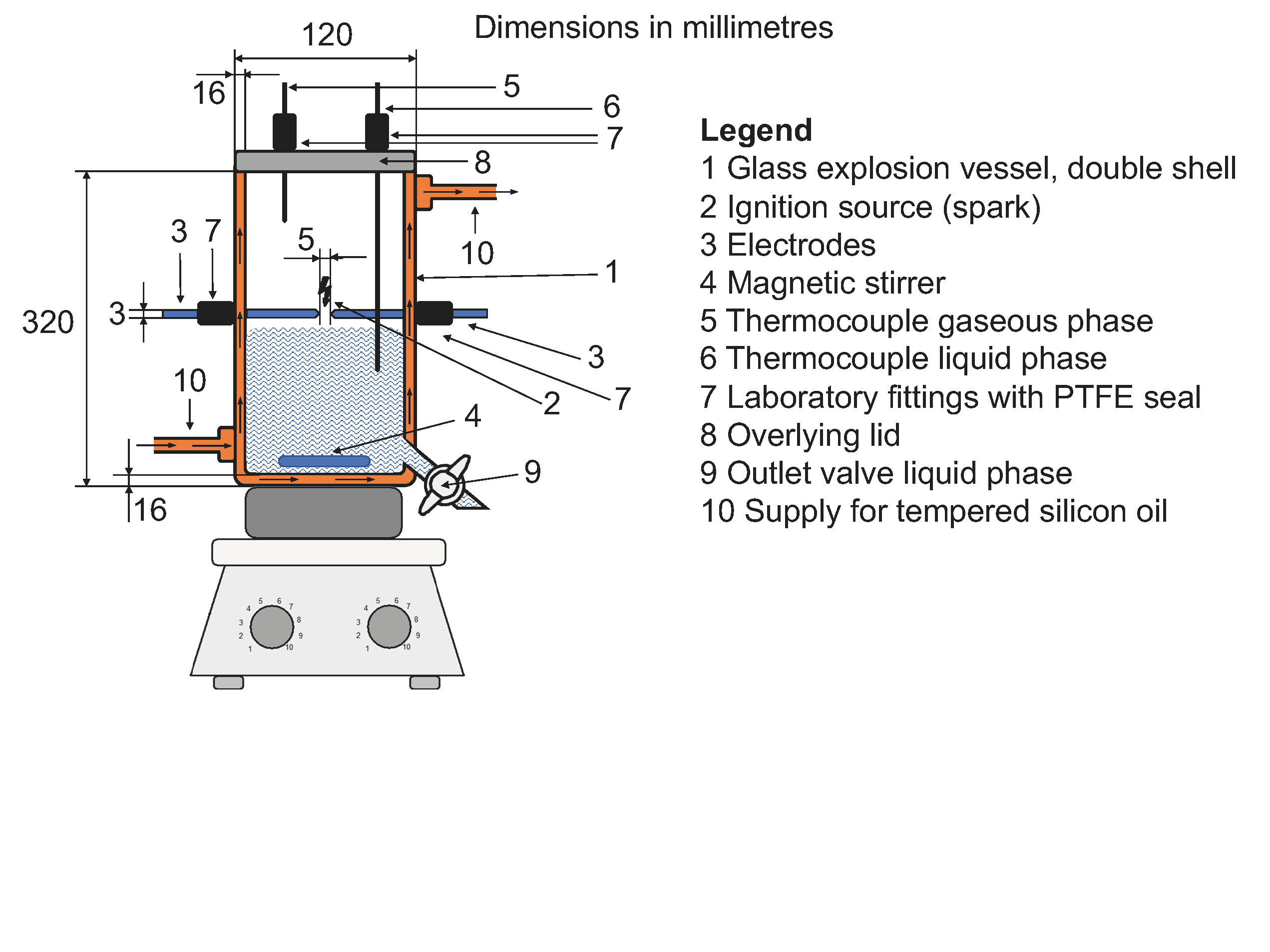
The temperatures of the sample and the vapor phase above it are recorded. The sample is stored for at least 0.5 h to allow sufficient time for equilibrium to be established. After the temperature stabilization time, an ignition spark (energy > 1 J) is used to check whether the gas atmosphere above the sample can be ignited.
The atmospheric pressure is recorded during the measurement.
The highest temperature at which no ignition occurs is used as the “lower explosion point at prevailing air pressure” LEP0 and must be converted to standard pressure (1013 hPa) using the air pressure prevailing at the time of measurement according to the following correction formula:
LEP = LEP0 + 0,025 • (1013 – p);
mit LEP = corrected lower explosion point in °C; LEP0 = measured lower explosion point in °C und p = air pressure in hPa





